
What is vapor pressure?
Vapor pressure: concepts and saturation
Vapor pressure is one of those quietly important quantities that runs through chemistry, physics, and everyday engineering without often being named in the headline. It describes what happens when a liquid sits in contact with its own vapor and the two phases settle into a steady arrangement. Get this concept wrong and you misread everything that follows: evaporation rates, boiling behaviour, condensation in cold pipes, even how a perfume disperses in a room.
What is vapor pressure and why does it matter?
Definition and characteristics of vapor pressure
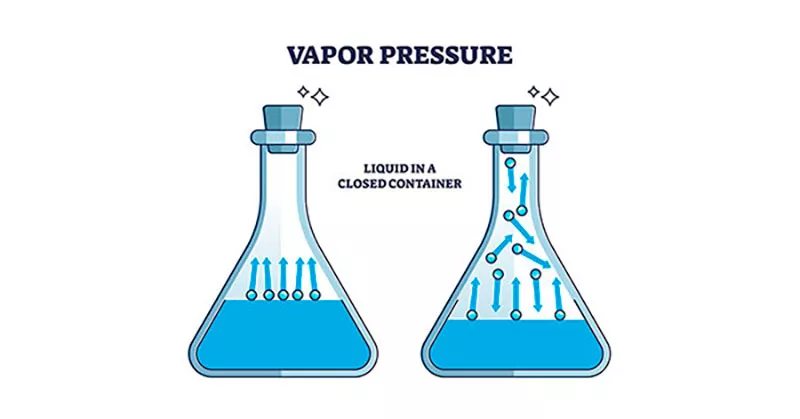
Inside a sealed container, a liquid will release molecules into the space above it. Some of those molecules drift back down and rejoin the liquid; others keep flying. When the rate of escape equals the rate of return, the system has reached equilibrium, and the pressure exerted by the gas phase at that point is what we call vapor pressure. The amount of liquid in the container does not change the figure — a teaspoon and a litre give the same reading at the same temperature. Temperature, on the other hand, changes everything. Heat the system, and the value rises sharply.
The importance of vapor pressure in chemical processes
Industrial chemistry leans on this number constantly. Distillation columns, drying tunnels, vacuum evaporators, even storage-tank design for volatile fuels — all of these run on calculations of how readily a liquid will turn into vapor at a given temperature. A liquid with a high vapor pressure evaporates eagerly, which is exactly what you want when separating an ethanol-water mixture and exactly what you do not want when storing petrol on a hot afternoon. The vapor pressure of water, taken alone, sits at the heart of meteorology, food drying, and humidity control inside semiconductor cleanrooms.
Comparison with other pressures: partial and atmospheric
Vapor pressure is often confused with two close relatives. Partial pressure refers to the share of total pressure contributed by a single component in a gas mixture — Dalton's territory. Atmospheric pressure is simply the weight of the air column pressing on whatever is below it, around 1 atm at sea level. Boiling, in fact, is the moment these worlds meet: a liquid boils when its vapor pressure climbs to match the atmospheric pressure pushing down on its surface. That is why water boils at 100 °C in Madrid but considerably lower on the summit of Mulhacén.
How is the vapor pressure of water measured?
Methods for measuring vapor pressure
Two approaches dominate the lab. An ebulliometer measures the boiling point of water at different external pressures, working backwards to derive vapor pressure from each result. Manometers, fitted to a sealed vessel, read the pressure built up by the vapor over the liquid directly. Both routes feed the same end: a reliable equilibrium value that thermodynamics and reaction kinetics can plug into their equations. Static, dynamic, isoteniscope, transpiration — there are more variants, each suited to a different temperature range or volatility regime.
How the boiling point influences the vapor pressure of water
Boiling point and vapor pressure are two readings of the same physical reality. Water boils at 100 °C only because, at that temperature, its vapor pressure has climbed to 1 atm. Lower the surrounding pressure and the boiling point drops with it; in a vacuum chamber, water can boil at room temperature without any heat applied at all. The relationship is not linear but exponential, which is why a small altitude gain knocks several degrees off the cooking temperature in a hill kitchen.
The relationship between evaporation and vapor pressure
Evaporation happens at any temperature, not just at the boiling point. Whenever the surface of a liquid is exposed to an undersaturated atmosphere, molecules with enough kinetic energy break free and join the gas phase. A liquid with a high vapor pressure evaporates faster because more molecules in its bulk reach the escape threshold at any given moment. Acetone evaporates from your skin within seconds; mineral oil will sit on a hot floor for weeks. The difference between them is, almost entirely, vapor pressure.
What is the relationship between vapor pressure and the boiling point?
How dynamic equilibrium influences the boiling point
The boiling point is not an arbitrary property of a substance — it is the temperature at which the dynamic balance between evaporation and condensation tips into something more violent. Until that point, both processes coexist at the surface of the liquid. At the boiling temperature, vapor begins to form within the bulk of the liquid itself, producing the bubbles we associate with the phenomenon. The vapor pressure has matched the surrounding atmospheric pressure, and the liquid no longer has any mechanical reason to stay liquid.
Variation in vapor pressure and its effect on the boiling point
Anyone who has tried to make a decent cup of tea on a mountain understands this in their bones. At high altitude the air is thinner, atmospheric pressure is lower, and water reaches the matching vapor pressure at a temperature considerably below 100 °C. The flip side applies in pressure cookers: raise the pressure inside the vessel and water can be pushed well past its sea-level boiling point, which is why beans cook in twenty minutes instead of four hours.
The role of dispersion forces
What holds molecules together in the liquid phase determines how easily they leave it. London dispersion forces, dipole-dipole interactions, hydrogen bonds — all of these work against vaporization. A substance held together by weak forces lets its molecules drift off without much persuasion, which pushes its vapor pressure up and its boiling point down. Diethyl ether, loosely bonded, slips into vapor at 35 °C; water, locked into a network of hydrogen bonds, holds out until the kettle hits 100.
Working out vapor pressure with Clausius and Clapeyron
What the equation actually says
Behind the formal-sounding name lies a fairly modest piece of algebra. Clausius-Clapeyron lets you take vapor pressure at one temperature and use it, together with the enthalpy of vaporization, to predict the value at any other temperature you care about. In its integrated form it links the natural logarithm of the pressure ratio at two temperatures to the enthalpy of vaporization, divided by the gas constant. Behind the algebra is a simple idea: the energy needed to push a molecule from liquid to gas does not change much over modest temperature ranges, so the vapor pressure rises in a predictable, exponential way as the temperature climbs.
Step by step calculation of vapor pressure
To apply the equation in practice, you need three pieces of information: the enthalpy of vaporization of the substance, a reference vapor pressure at a known temperature, and the temperature you want to convert to. Plug those into the integrated form, solve for the unknown pressure, and the answer falls out. Tabulated values for water, ethanol, benzene, and most common refrigerants are widely available, which is why the equation is so heavily used in process design.
Practical uses of the equation
Refrigeration cycles depend on the relation. Choosing a refrigerant means picking a fluid whose vapor pressure curve passes through the right temperatures at the right pressures for the duty cycle. Meteorologists use the same equation to model how saturation pressure of water vapor changes with altitude and temperature, which feeds directly into cloud formation and dew point calculations. From a freezer in a corner shop to a satellite weather forecast, Clausius and Clapeyron are doing the heavy lifting.
What does Raoult's law say about vapor pressure?
Effects of liquid mixtures on vapor pressure
When two or more liquids are mixed, the vapor pressure of the mixture is no longer that of either pure component. Raoult's law states that, in an ideal solution, the partial vapor pressure of each component equals its vapor pressure as a pure substance multiplied by its mole fraction in the liquid. The total vapor pressure above the mixture is the sum of those partial contributions. From this single relation flow most of the colligative properties students meet in their first physical chemistry course: boiling-point elevation, freezing-point depression, osmotic pressure.
Vapor pressure in solutions: theory and examples
Take a solution of ethanol and water at 25 °C. Pure ethanol has a vapor pressure of about 7.9 kPa at that temperature; pure water, around 3.2 kPa. A 50:50 mole-fraction mixture, if it behaved ideally, would generate a total vapor pressure equal to half of each value added together — close to 5.6 kPa. Real ethanol-water mixtures deviate, because hydrogen bonding between unlike molecules differs from bonding between like ones, but the calculation gives the right order of magnitude and a starting point for refinement.
Limitations and considerations of Raoult's law
Raoult's law works cleanly for chemically similar species — benzene and toluene, for instance, behave almost ideally across the full composition range. Things get messier when intermolecular forces between unlike molecules differ markedly from those within each pure component. Aqueous solutions of strong electrolytes, mixtures of polar and non-polar liquids, and anything involving association or dissociation all show measurable deviations. For these cases, activity coefficients are introduced as correction factors, and engineers reach for models such as NRTL, Wilson, or UNIQUAC instead of the bare law.
